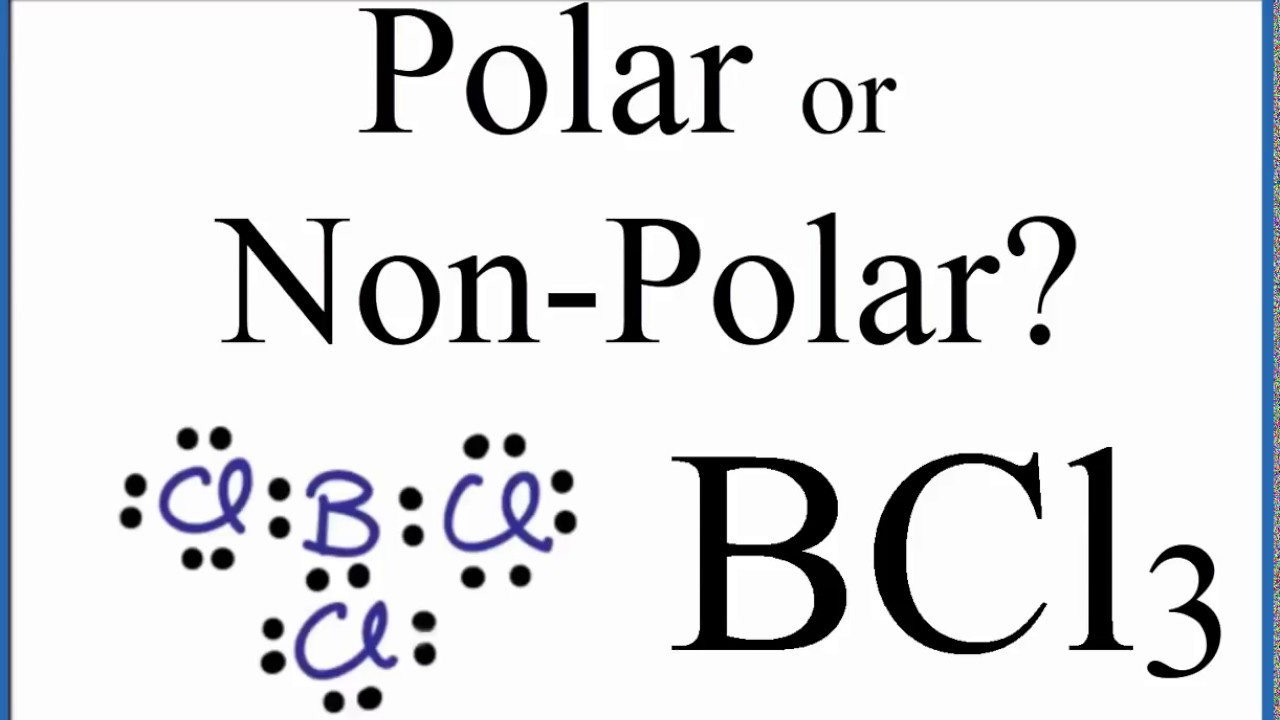
Why BCl3 has zero dipole moment?ī and Cl atoms have different values in terms of electronegativities, hence chlorine (E.N. 
The central atom is distributing symmetric charge throughout the molecule and thus is Nonpolar!įor more detailed information, you must read an article written on lewis structure of BCl3. If we look at the Boron Trichloride’s molecular geometry, it is Trigonal Planar with a bond angle of 120 degrees. No such dipole exists in BCl3 and thus its dipole moment is zero and it is non-polar. To be polar, a molecule must have an asymmetrical shapeshift in electron density such that an electrical dipole is formed. Note that, Boron doesn’t fully complete its octet, but is still able to create a non-polar molecule. This indicates that the pull of these atoms is perfectly balanced out in this compound. This molecule is forming a triangular shape with an angle of 120 degrees splitting up the chlorine atoms with the same angle. This causes the molecular configuration for the chlorine atoms to be a part of perfect triangular shape. This happens due to BCl3 being an electron-deficient molecule that easily accepts a pair of the electron from water in Hydrolysis.Ĭonclusion Molecular Geometry and Bond Angle of BCl3Ĭentrally located Boron of this molecule has three valence electrons, which balances out the three chlorines. When Boron Trichloride reacts with water, it produces boric acid and hydrogen chloride as a substitute compound. If kept in prolonged exposure to fire or extreme heat, the containers may rupture itself or even burst out like a rocket violently.īCl3 is mainly used as a catalyst in chemical manufacture, and soldering fluxes. It is corrosive to metals, tissues, and considered toxic in nature. As a result, the dipole moment of each B-Cl bond is canceled out by each other making the net dipole moment zero and the entire molecule nonpolar in nature.įumes originating from the chemical compound BCl3 can easily irritate the eyes, along with the mucous membranes. The B-Cl bond itself is polar because of the difference in electronegativity of Boron(2.04) and Chlorine(3.16) atoms and all three B-Cl bonds lie at 120 degrees to each other. So, is BCl3 polar or nonpolar? Boron Trichloride or BCl3 is a nonpolar compound because of its symmetrical structure ie Trigonal Planar. 
So, in this article, I will answer this and cover the fundamentals of the polarity of the BCl3 compound. One common question is generally asked about its polarity. This chemical compound is a colorless gas with a pungent odor and acts as a reagent in organic synthesis. Boron Trichloride is an inorganic compound with the molecular formula (BCl₃).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
